|
Our lab has been awarded two grants from the Italian Ministry for University and Research (MUR) as part of the "Progetti di Rilevante Interesse Nazionale" (PRIN) program. As part of the PRIN 2022 PNRR call, our project entitled: "From stem cells to algae and back: circular biotechnology for cellular agriculture and biomass production" was ranked 4th overall in the LS9 scientific sector, and 1st among the "Under 40" category. This project is led by our lab and involves an exciting interdisciplinary collaboration with Alberta Pinnola's plant physiology group at the University of Pavia. The total budget is € 230,861. In this proposal we tackle a pivotal challenge towards the pursuit of a sustainable and circular-economy-based future: how can we provide food within planetary limits for an expanding population with a growing appetite for meat and fish? This is our first research grant in the cellular agricolture space, which we are super excited about. As part of the PRIN 2022 call, our project entitled: "Decoding and leveraging the molecular determinants of myogenic fate through integrative genomics and cell engineering" received a score of 83/100 in the LS2 scientific sector. The project is led by Davide Cacchiarelli's integrative genomics lab at TIGEM and involves the collaboration of our group as well as Annamaria Carissimo's bioinformatics team at CNR. The total budget is € 199,628. The overarching goal of this proposal is to first uncover and then exploit the gene regulatory networks behind cell fate conversion into muscle. The knowledge generated by this proposal will enable large scale, cost-effective generation of mature muscle cells for various applications. These two grants support our emerging lines of research in cardiac regeneration and cellular agriculture. Both grants have a two year duration and will start in the Fall of 2023. We are looking for postdocs motivated to develop these exciting lines of research, so please spread the word and reach out if interested.
0 Comments
This epic tale begins some 6 years ago. The team of Chuck Murry at the University of Washington (UW) had spent almost two decades refining methods to grow large number of cardiomyocytes differentiated from human pluripotent stem cells (hPSC-CMs), and honing in the best way to deliver these to the injured myocardium so that they would survive, grow in size, and mature to become functional portions of a remuscolarized cardiac wall. The goal of moving to clinical trials had felt exceedingly close, until in the early '2010s studies in large animal models came as a wet blanket by revealing the high prevalence of engraftment-associated arrhythmias (EAs). This was a major roadblock that required an "all hands on deck" approach. The predominant hypothesis was that the immaturity of hPSC-CMs was to blame, leading them to behave as an ectopic "pacemaker". Many approaches had been tested to improve the maturity of hPSC-CMs, such as mechanical, hormonal, or metabolic stimulation. All had provided some important advances, but none had yet fully succeeded.
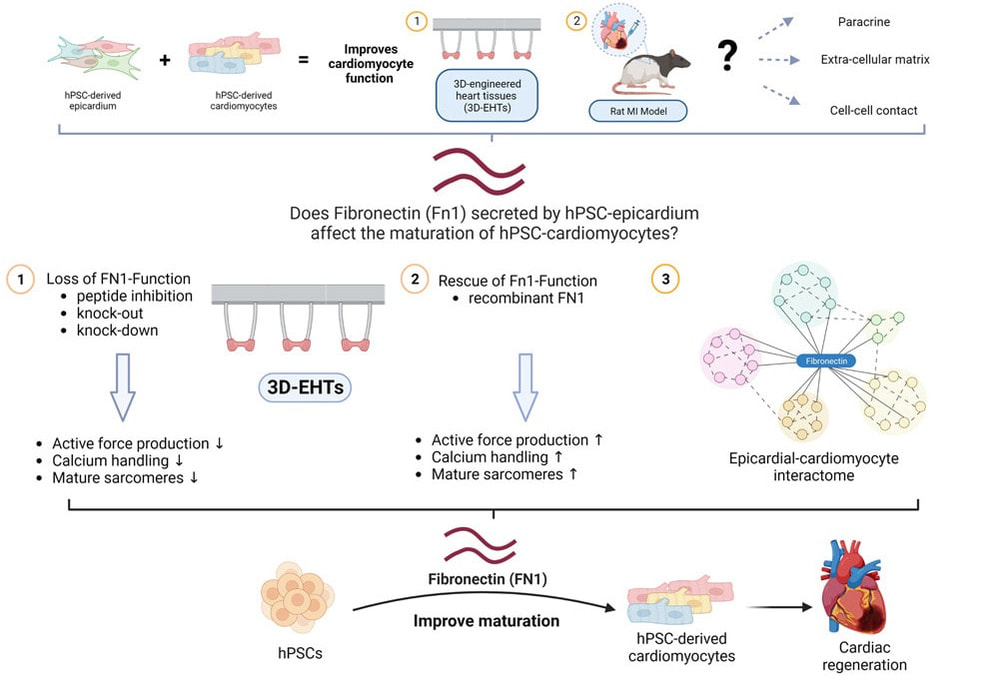
Desperate times call for desperate measures, and so Chuck decided it was time to try a drastic approach to eliminate the problem at its root: modify the genome to "educate" adolescent hPSC-CMs to behave like adults. He established a "Strikeforce" whose goal was to identify the right targets, edit them in or out of the genome of hPSCs, and test the resulting hPSC-CMs for their ability to spontaneously beat in vitro, a possible surrogate of their arrhythmogenic properties in vivo. When I had joined Chuck's lab as a postdoc a couple of years prior it was for the 4D Nucleome project, a basic science effort to discover the functional changes in 3D chromatin structure in cardiac development and disease. Since then, I had grown increasingly interested in the efforts of the Heart Regeneration Project (HRP), but I could not see how to contribute given my background in gene regulation and genome editing. Until one day, during a long walk along Lake Union, Chuck asked me if I wanted to lead the Strikeforce. It was a fork in the road I took eagerly: the rest, as they say, is history. The MEDUSA project was the most ambitious I have ever been part of; it thought me patience, resilience, leadership, and the true meaning of teamwork. Besides the Strikeforce's members, first and foremost lead author Silvia Marchianò, some 25 scientists across academia and industry were all pivotal to produce cells in large scale, transplant them, and study the resulting animals and tissues. What was supposed to be a quick "in and out" operation turned into a gruelling war of attrition with an enemy that proved crafty and full of resources. After generating some 12 gene edited lines, we finally identified a combination of 4 genome edits able to *finally* prevent spontaneous beating in hPSC-CMs in vitro, yet maintain their ability to respond to an electrical signal. In agreement with our initial hypothesis, their transplantation did not result in EA. Following the footsteps of Perseus, we had finally slayed the MEDUSA. I left the UW to start my lab in Italy two years ago almost exactly, and since then the study continued, marching through a though but fair peer review, among other things confirming our initial safety findings also when a larger dose of hPSC-CMs were transplanted. I cannot thank Chuck enough for his steadfast support of the study even after the many failures at the beginning. The Strikeforce and HRP as a whole are the inspiration for the program I am working hard to establish, with different goals, also on our side of the pond. The mythos does not end here: there are still several monsters to be slain on the path towards remuscolarizing the heart of patients in a safe, efficient, and scalable manner. Still, today we stop and celebrate that we are, at last, a big step closer. Alessandro Bertero Transatlantic collaboration identifies key molecule for cardiac bioengineering (and regeneration?)3/30/2023 Our friends and collaborators in the Sinha lab in Cambridge published an exciting paper in Stem Cell Reports that reports the key role of an extracellular matrix component, fibronectin, in the maturation of cardiomyocytes seeded in 3D scaffolds together with epicardial cells (forming so-called 3D Engineered Heart Tissues, 3D-EHTs). The study involved a transatlantic collaboration with the Murry lab in Seattle, WA. We were glad to contribute to the genome editing experiments that led to the formal demonstration that fibronectin is required for the pro-maturation activity of epicardial dells.
A previous study from the same collaborative group had identified they key ability of epicardial cells to promote cardiomyocyte maturation both in vitro and in vivo, but the mechanism remained unclear. This study identifies fibronecting as being both sufficient and necessary for this process in vitro, which opens the door to simplified and more reproducible bioengineering applications compared to adding epicardial cells.
Fibronectin may prove key to also boost cardiac remuscolarization in vivo. Indeed, the immaturity of human pluripotent-derived cardiomyocytes (hPSC-CMs) is thought to be a key driver of arrhythmic complications following their transplantation: what if a pinch of fibronectin in transplanted hPSC-CMs improved their safe engraftment and function?There promises to be a third episode of this saga.. |